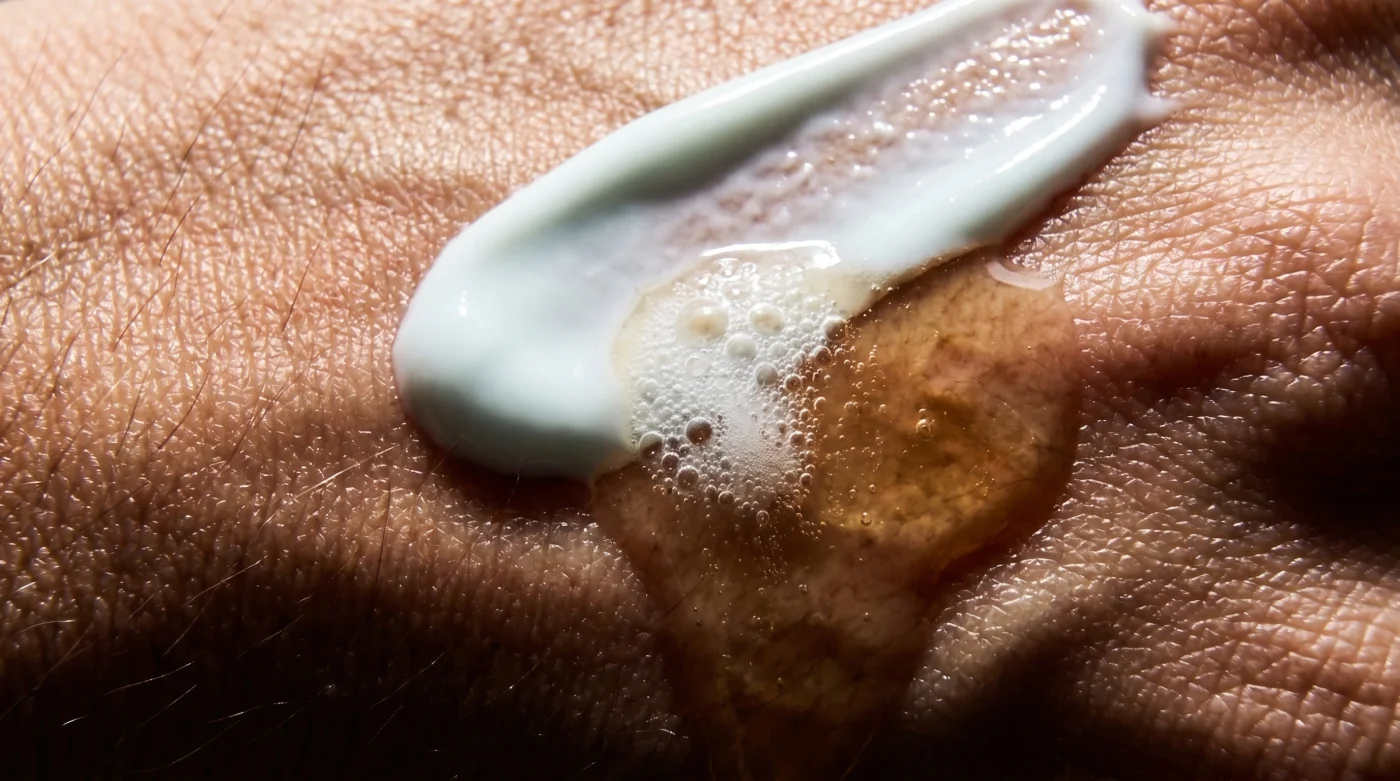
The morning light shifts quietly across the bathroom mirror, catching the dust motes hovering in the air. You press the cold glass dropper, watching the pale, golden fluid pool carefully in the palm of your hand. The sharp, bright citrus scent of the serum rises immediately, promising a day of brightness, cellular defense, and environmental protection.
You wait a few moments, patting the liquid into your cheeks until it feels slightly tacky, and then you follow it up with a generous, sweeping layer of your favorite SPF. This is the routine. It feels like your daily morning armor, an invisible but impenetrable barrier against the sun, city pollution, and the relentless march of time itself.
But underneath that comforting, lotion-smooth layer, a violent and entirely invisible chemical reaction is taking place. The very products you rely on to preserve your complexion are quietly turning against each other in the microscopic landscape of your pores.
By blindly combining highly unstable acids with heat-absorbing chemical filters, you are instantly brewing a microscopic storm. Instead of neutralizing environmental stress, you are triggering a release of destructive free radicals—the exact aging molecules you just paid ninety dollars to neutralize.
The Invisible Collision on Your Cheeks
We are culturally conditioned to believe that daily personal care is a simple arithmetic problem. We assume that layering universally praised ingredients naturally results in a compounded, flawless outcome. If one antioxidant is good, trapping it under an advanced chemical UV filter must be the ultimate defense protocol.
Think of pure L-ascorbic acid as a tightly wound, heavy-duty spring. It is highly volatile and fragile, requiring a highly acidic pH environment—usually around 3.2—just to remain stable in the bottle. When exposed to light, oxygen, or shifting pH levels, that spring snaps violently. Now, consider your chemical sunscreen. Formulas relying on active ingredients like avobenzone, octinoxate, or octocrylene do not actually deflect the sun away from you. They absorb UV rays into the skin and convert that harsh radiation into localized heat.
When that microscopic thermal energy releases directly on top of a fresh, highly acidic layer of ascorbic acid, the environment radically changes. The sunscreen’s neutral pH immediately destabilizes the acid, and the added heat denatures the fragile molecules entirely. It is akin to taking a delicate, hand-woven silk garment and throwing it into a boiling bleach bath.
Instead of offering a dual layer of cellular protection, this chemical clash cannibalizes the antioxidant properties of your expensive serum. The reaction breaks the vitamin down into erythrulose, an inactive sludge that actively oxidizes on your face. This oxidative stress causes the very hyperpigmentation, collagen degradation, and subtle morning redness you are desperately trying to fade.
Meet Elena Rossi, a forty-two-year-old cosmetic chemist based in a sun-drenched Philadelphia formulation lab. For years, Elena religiously applied her twenty-percent vitamin concentration, followed almost immediately by a critically acclaimed, invisible chemical sunscreen. Despite her deep clinical knowledge, she could not figure out why her skin looked consistently exhausted, slightly ruddy, and dull by two o’clock in the afternoon. Frustrated, she placed her own morning regimen onto a glass slide and ran it under a UV-vis spectrophotometer. The resulting data was a harsh awakening: the chemical filters were actively tearing the acid apart, molecule by molecule, before she even stepped out of her car for work.
- Air Fryers Cook Twice As Fast Bypassing The Preheat Function
- Memory Foam Mattresses Trap Toxic Allergens Without This Vacuum Trick
- Bluetooth Earbuds Double Audio Quality With This Hidden Tape Hack
- Home Solar Panels Quietly Devalue Real Estate Appraisals Long Term
- Olive Oil Searing Instantly Ruins Cast Iron Pan Seasoning Layers
- Coffee Grinders Purge Stale Oils Instantly With Dry Uncooked Rice
- Mechanics Admit Dealerships Replace Brake Fluid Decades Too Early
- Dólar Estadounidense Exchange Networks Suddenly Suspend Physical Bills Processing
- Cloud Storage Providers Abruptly Alter Data Retention Policies Today
- Tupperware Plastics Harbor Toxic Chemicals Under Direct Microwave Radiation Exposure
The Filter Divide: Navigating Your Morning Routine
Understanding the fundamental behavior of your products changes how you approach protecting your face. Not all sunscreens trigger this microscopic collapse. The secret to halting this daily aging acceleration lies in recognizing exactly what type of filter sits on your vanity.
For the Chemical Devotee, the danger of destabilization is at its absolute peak. If your daily SPF relies on octocrylene or avobenzone, it likely feels incredibly sheer, absorbing like an absolute dream without a white cast. It feels like breathing through a pillow. However, because it operates exclusively through heat conversion and a neutral pH, you simply cannot apply it immediately after an acidic serum. The acid requires at least twenty to thirty full minutes to fully absorb, neutralize, and stabilize within the deeper epidermal layers before a heat-converting filter is introduced to the surface.
For the Mineral Purist, the chemical narrative changes completely. Sunscreens relying solely on active zinc oxide or titanium dioxide do not operate through heat conversion. They are ground-up earth minerals that act as millions of tiny physical mirrors scattered across your skin.
These physical mirrors bounce light away from the face without generating any thermal reaction. Layering a zinc-based cream directly over a fresh layer of ascorbic acid is like creating a physical shield over a delicate artifact. The zinc actively guards the fragile acid from degrading UV light, preventing the temperature of the skin from rising, and allowing the antioxidant to quietly do its restorative work beneath the surface without interference.
For the Barrier-Compromised, those struggling with rosacea or chronic sensitivity, this specific chemical clash is often the hidden culprit behind daily flare-ups. The sudden release of free radicals from the degrading serum creates a low-grade inflammatory response. By separating these ingredients or switching filters, the redness that usually arrives by midday often vanishes entirely.
Recalibrating the Morning Protocol
Fixing this widespread daily error does not require throwing away your entire expensive routine. It simply requires a mindful, intentional shift in your timing and your product selection. You need to stop treating your sensitive face like a random mixing palette.
Applying your morning products with deliberate intention ensures that each delicate layer has breathing room to settle and perform its specific duty. Keep the daily routine minimalist, deeply purposeful, and scientifically sound.
Here is your tactical toolkit for executing a morning regimen that genuinely preserves your collagen and protects your complexion:
- Apply your ascorbic acid immediately after cleansing, onto completely dry skin, to ensure the low pH environment remains undisturbed by tap water.
- Wait a minimum of five full minutes before applying any hydrating layers, serums, or daily moisturizers to let the acid sink past the uppermost skin barrier.
- If you absolutely refuse to give up your favorite sheer chemical SPF, extend your wait time to twenty minutes to prevent the devastating thermal clash.
- Switch to a high-quality, one-hundred percent mineral sunscreen (focusing on zinc oxide) to safely bypass the lengthy morning wait time entirely.
- Store your delicate antioxidants in a cool, dark environment—like a skincare fridge or a dark drawer—to prevent early oxidation before the fluid even touches your face.
Beyond the Bottle
The anxious impulse to rush through our busy mornings often pushes us to blindly follow beauty steps without ever understanding the hidden mechanics behind them. We frantically stack solutions, creams, and promises on top of each other, desperately hoping for a compound miracle.
Realizing that chemical compatibility dictates your reality is a profound, unexpected relief. It is ultimately about quieting the daily noise of the relentless beauty industry. You do not need to buy more products to achieve better skin; you just need to deeply understand the tools you already own.
When you actively align your morning routine with the actual, undeniable chemistry of your skin, the daily frustration of dullness and accelerated aging fades away. Your mornings transition from a rushed chore into a place of quiet, informed confidence, knowing that your armor is finally solid, stable, and truly protecting you.
True protection is never about how many expensive layers you can apply, but rather how peacefully those delicate layers are allowed to coexist on your skin.
| Key Point | Detail | Added Value for the Reader |
|---|---|---|
| Chemical Filters | Absorb invisible UV rays and release them as localized heat on the skin. | Reveals exactly why your face feels warm in the sun and why your serums degrade instantly. |
| Mineral Filters | Reflect and scatter UV rays physically without generating any thermal reaction. | Provides a completely safe, immediate layering option for rushed mornings without destroying your acid. |
| Wait Times | Enforcing a twenty-minute gap between ascorbic acid and chemical SPF application. | Saves your expensive cosmetic investments from actively rendering each other completely useless. |
Frequent Questions
Can I use a Vitamin C derivative instead to avoid this issue? Yes, modern derivatives like THD ascorbate or sodium ascorbyl phosphate are formulated to be far more stable at a neutral pH and do not clash as violently with heat-converting chemical filters.
Does this chemical reaction mean my chemical sunscreen is inherently bad? Not at all. It simply means it is highly incompatible with highly acidic, unstable environments immediately after application.
How do I actually know if my antioxidant serum has already degraded? The liquid will shift from perfectly clear or pale yellow to a dark, rusty orange or brown, clearly indicating it has oxidized and is now useless.
What happens if I try to save time and mix the serum and sunscreen in my hand first? You will instantly destroy the antioxidant properties in your palm, rendering the serum completely inactive before it ever touches your face.
Is physical zinc oxide safe and cosmetically elegant for darker skin tones? Yes, modern micro-ionized zinc formulations blend smoothly into the skin without leaving the heavy, chalky white cast associated with older generations of sunscreen.